5.2 Indirect Heating of Thermal Fluids
Many operations, requiring energy delivery to a liquid-phase (and/or fluid) stream, require a physical barrier between the fossil combustion products (energy release) and the process stream.
The barrier reduces heat transfer efficiency, but is often necessary. This is shown in schematic in Figure 5.4. Traditional systems depend upon heat delivery via heat exchangers, fire-tube schemes (heating coils, or multi-pass fluid heaters) and other methods. Many of these systems use flame- induced, radiant-based heating to rapidly deliver well over 50% of the required energy.
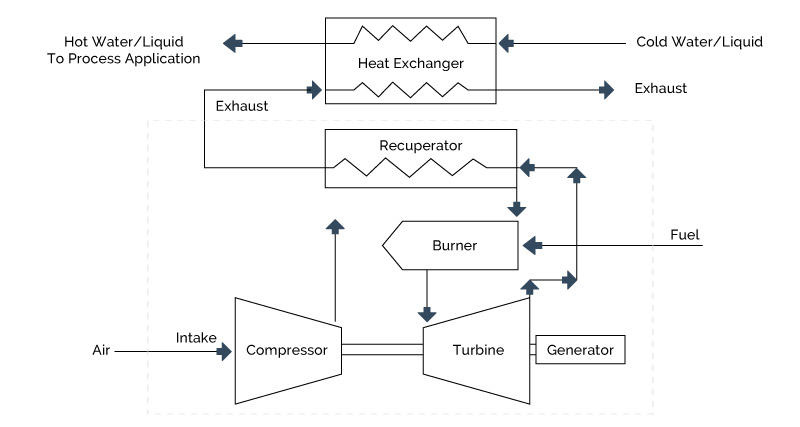
Figure 5-4: Indirect Fluid Heater System
5.2.1 Process Uses
Processes that use high (and/or variable) pressure systems, separation/purification operations, multiphase operations, systems impeded by oxidation (or other possibly reactive/degrading components of combustion), and strictly maintained closed-loop systems are common boundaries to direct heating of process streams. More specific operations include:
- Purification, recovery, and separations.
- Chemicals and refinery distillation towers, reboilers, and flash evaporators for polymer processing, slurry separations/purification, brine treatment, etc.
- Pressurized process streams (in chemical reactors, etc.).
- Processes and products sensitive to oxidation, other reaction-driven degradation, and/or general fouling (in chemical, food, pharmaceutical processing, etc.).
- Vat or batch systems maintaining a heated fluid (such as paint/dye blenders, food deep fryers, refinery-bottoms
storage and subsequent processing, reactor/fermentation vessels, crystallizers, etc.). - Thermal fluid, closed-loop-heating systems for processes requiring especially high and smoothly controlled
temperature profiles.- Systems requiring high temperatures over large areas such as calcium chloride crystallizers.
- Pipe line tracing.
- Distillation and reactor feed lines whereby preheating feed components simplifies the energy delivery and/or chemistry complexity of any downstream operation.
- Heat tracing viscous material (crude, confectionery, polymer melts, etc.) pipelines to reduce electric-driven pumping rquirements.
- Tool heating (including plastics/rubber extruders, molds), paper mill platens and rollers, metal fabrication equipment, laminate setting, and others.
- General polymer processing. Polymer processing plants may require high temperature (> 400 °F) energy delivery to several unit operations because of high “pure” polymer melting points (maintained for extrusion, molding, etc.), and endothermic and/or equilibrium limited reactions (whereby light byproducts, often water, must be continuously evaporated and removed for effective/efficient reactor output). Polyester and Nylon are good examples of major international commodities often utilizing thermal fluids systems throughout their production cycle.
